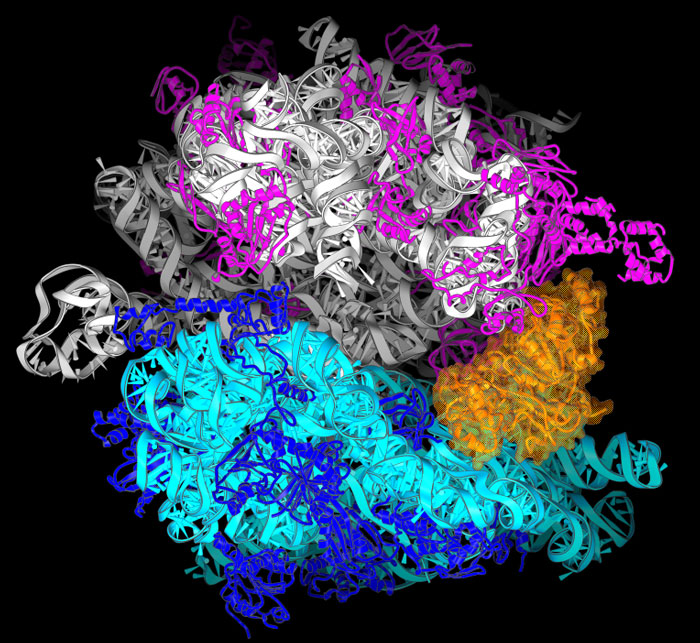
Crystal Structure of Ribosome with Release Factor RF3
Members of the Noller lab recently published a 3.3 Å resolution structure of a complex containing the ribosomal release factor (RF3) locked in its GTP-bound state (mimicked using GDPNP) in association with the *E. coli* 70s ribosome. Crystallographic data for this project was collected at the SIBYLS beamline MX station.
>
The class II release factor RF3 is a GTPase related to elongation factor EF-G, which catalyzes release of class I release factors RF1 and RF2 from the ribosome after termination of protein synthesis. The 3.3 Å crystal structure of the RF3·GDPNP·ribosome complex provides a high-resolution description of interactions and structural rearrangements that occur when binding of this translational GTPase induces large-scale rotational movements in the ribosome. RF3 induces a 7° rotation of the body and 14° rotation of the head of the 30S ribosomal subunit, and itself undergoes inter- and intradomain conformational rearrangements. We suggest that ordering of critical elements of switch loop I and the P loop, which help to form the GTPase catalytic site, are caused by interactions between the G domain of RF3 and the sarcin-ricin loop of 23S rRNA. The rotational movements in the ribosome induced by RF3, and its distinctly different binding orientation to the sarcin-ricin loop of 23S rRNA, raise interesting implications for the mechanism of action of EF-G in translocation.