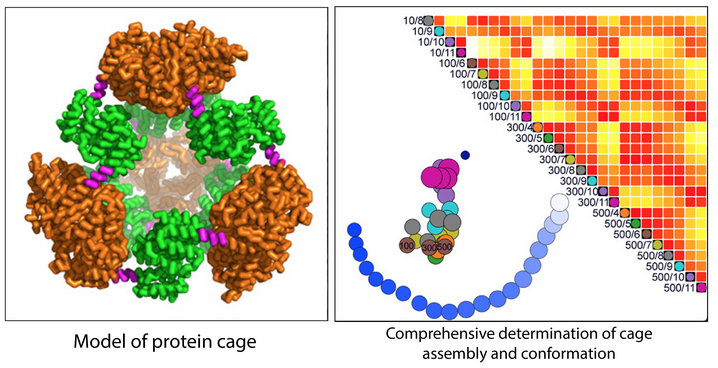
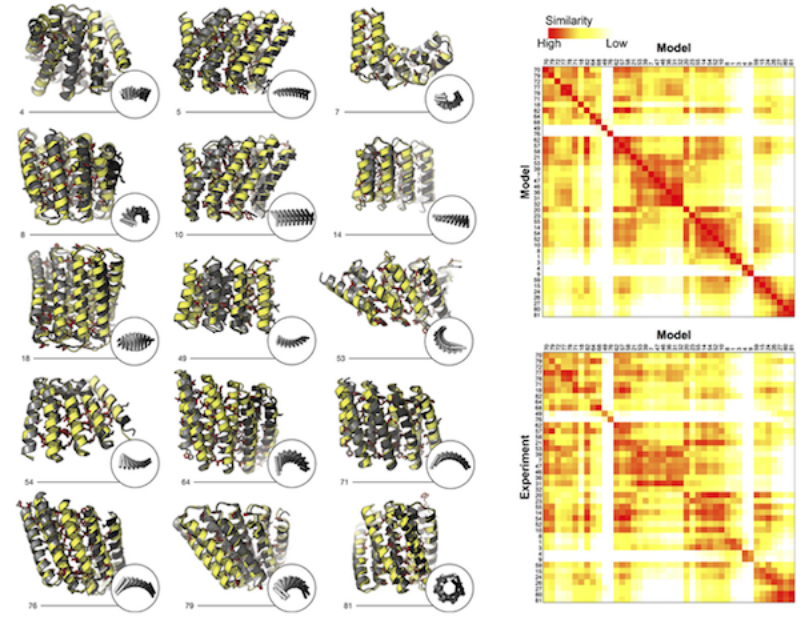
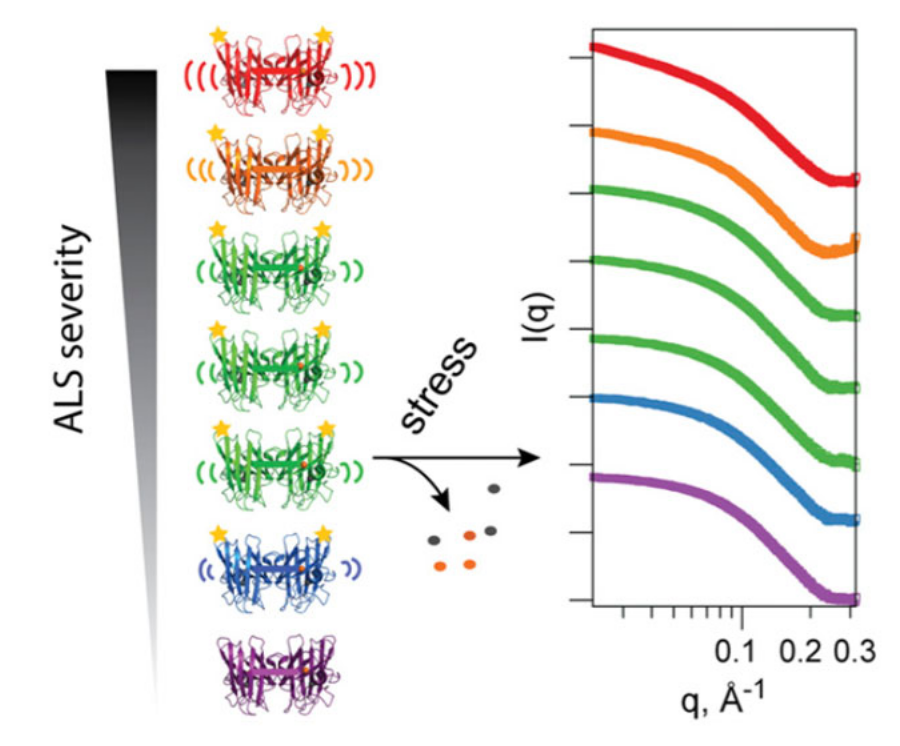
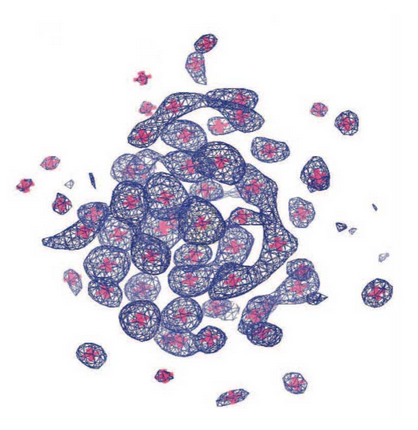
Designing and defining dynamic protein cage nanoassemblies in solution
Central challenges in the design of large and dynamic macromolecular assemblies for synthetic biology lie in developing effective methods for testing design strategies and their outcomes, including comprehensive assessments of solution behavior. The authors of this paper created and validated an advanced design of a 600-kDa protein homododecamer that self-assembles into a symmetric tetrahedral cage.…
Read more